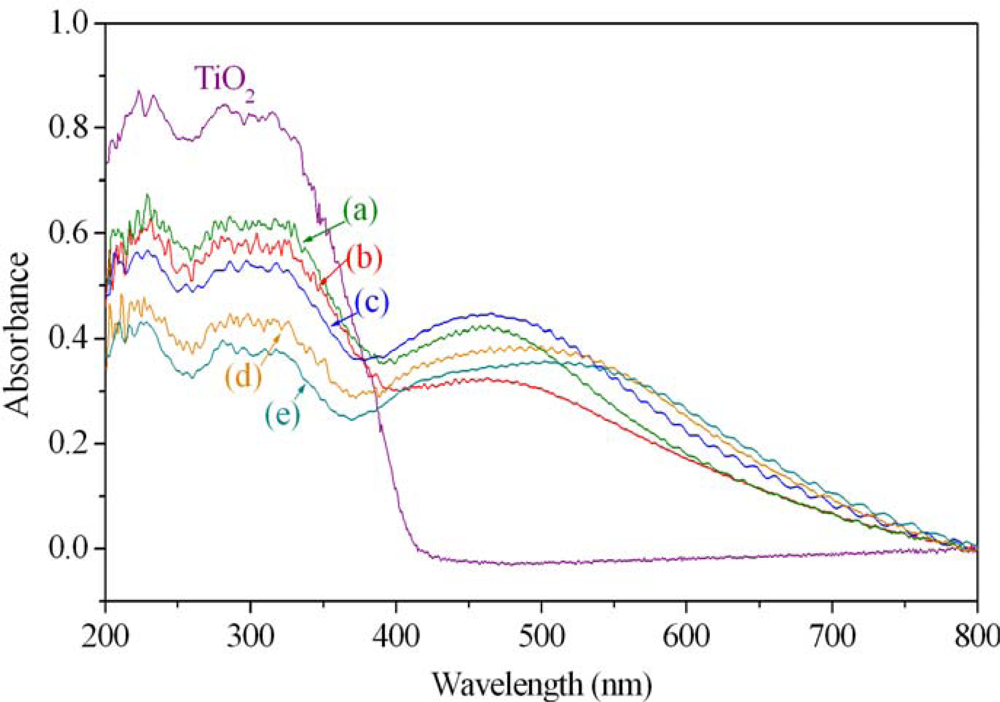
Nevertheless, the practical applications of TiO 2 have a major drawback, namely, they are active only in UV light ( 430 nm) can be attained by doping the TiO 2 network with non-metals, lanthanide ions, transitional metal ions, noble metals or metallic oxides. Titanium dioxide catalysts proved to be better than the other catalysts studied in literature (ZnO, SnO 2, WO 3, CdS) because of their superior redox ability and photoelectric properties, the long-term stability, the nontoxicity, and the low cost. Since 1972, when Fujishima and Honda published their seminal work, much work has been focused on investigating the photocatalytic properties of TiO 2. Over the last years, titania nanomaterials have attracted a lot of attention as they have found numerous applications in the field of dye-sensitized solar cells, Li-ion batteries, sensors, photodynamic cancer therapy or in biomaterials. These findings could have implications in the development of new nanocatalysts. Also, it was established that such hybrid films can be reused at least for five cycles, without losing too much of the photocatalytic efficiency (ca. The degradation of the mentioned model pollutants varied between 71% and 100% (after 250 min of irradiation) depending on the composition of the hybrid film tested and the light applied (UV–visible light). The best results were obtained for the films with TiO 2/Fe 2O 3 or TiO 2/SiO 2/Fe 2O 3 NPs. The catalytic activity of these hybrid films was tested for the photodegradation of phenol, hydroquinone and dopamine in aqueous solution under UV or visible-light irradiation. The mean size (10–30 nm) and the crystallinity of the NPs varied as a function of the inorganic constituent. The physicochemical characterization of nanoparticles and hybrid polymeric composites with 10 wt % NPs (S1–S4) was realized through XRD, TEM and FTIR analyses. Moreover, the as-prepared TiO2 nanoparticles showed a high PL intensity due to an increase in the recombination rate of photogenerated electrons and holes under UV light irradiation.Polymer nanocomposites containing titanium oxide nanoparticles (TiO 2 NPs) combined with other inorganic components (Si–O–Si or/and γ-Fe 2O 3) were prepared by the dispersion of premade NPs (nanocrystalline TiO 2, TiO 2/SiO 2, TiO 2/Fe 2O 3, TiO 2/SiO 2/Fe 2O 3) within a photopolymerizable urethane dimethacrylate (polytetrahydrofuran-urethane dimethacrylate, PTHF-UDMA). A blue shift was observed in UV-vis absorption spectra due to the quantum size effect of TiO2 nanoparticles. After subsequent hydrolysis of the anchored Ti complexes and calcination of the amorphous TiO2, anatase TiO2 nanocrystals with spherical shape and uniform particle diameter of about 6 nm were formed. It was found that SBA-15 contained abundant silanol groups after removal of triblock copolymers by ethanol extraction and could easily adsorb a great number of titanium alkoxide via chemisorption. The as-synthesized samples were characterized with Fourier transform infrared spectra (FTIR), Raman spectroscopy, diffuse reflectance UV-visible spectroscopy (UV-vis), powder X-ray diffraction (XRD), small-angle X-ray diffraction (SAXRD), nitrogen adsorption, transmission electron microscopy (TEM) and photoluminescence spectra (PL). Highly dispersed TiO2 nanoparticles were successfully synthesized by a wet impregnation method using SBA-15 as hard template for confining the growth of TiO2 nanocrystals, and then calcined at 550 degrees C in muffle furnace for 2 h.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
